|
Sensor information focuses on the GPU, HDD, CPU, RAM and others. They may also monitor hard disk temperatures for each core to prevent overheating. HWiNFO64 also shows sensor-related data to help users figure out the best course of action when it comes to upgrading hardware components, for example. Analyze sensor information to oversee the PC's health status 
You can create reports with a preferred file type between LOG, CSV, XML, HTML, MHTML and TXT with any selected computer areas or everything. attributes, audio driver data, along with network MAC address and maximum link speed. Investigate device information and create detailed reportsįor instance, it is possible to find out the standard and enhanced features for the processor, BIOS version, internal graphics support, each memory row's serial number and refresh rate, processor clock, graphics driver information, monitor width and height and supported video modes, drive S.M.A.R.T. They are organized in a tree view where you can access information related to the central processor, motherboard, memory, bus, video adapter, monitor, drives audio, network and ports. You may also enter the settings panel from this area. At startup it is possible to set HWiNFO64 to show only sensors or only the summary. The setup procedure is a fast and easy task that does not require special attention. In January 2022, the FDA also expanded the use of remdesivir to treat non-hospitalized patients with mild-to-moderate COVID-19 who are at high risk for progression to severe disease.HWiNFO64 is a powerful system information utility that gathers important data on the computer's hardware components, such as sensor information and S.M.A.R.T. 
On December 21, 2021, the indication expanded to include the treatment of COVID-19 in adults who do not require supplemental oxygen and who are at increased risk of progressing to severe COVID-19. Remdesivir was also approved by the European Commission on Jfor the treatment of COVID-19 in adults and adolescents with pneumonia requiring supplemental oxygen. Remdesivir in combination with baricitinib for the treatment of COVID-19, was granted an FDA Emergency Use Authorization on November 19, 2020. Remdesivir is currently marketed under the trademark name VEKLURY® by Gilead Sciences Inc. The FDA subsequently granted full approval for remdesivir as a COVID-19 treatment on October 22, 2020, while simultaneously updating the EUA to cover those patients not included under the approved indication. Remdesivir was confirmed as a non-obligate chain terminator of RdRp from SARS-CoV-2 and the related SARS-CoV and MERS-CoV, and has been investigated in multiple COVID-19 clinical trials.īased on aggregate data, remdesivir was granted an FDA Emergency Use Authorization (EUA) on May 1st, 2020. Remdesivir activity against the Coronaviridae family was first demonstrated in 2017, leading to considerable interest in remdesivir as a possible treatment for COVID-19. Broad antiviral activity of remdesivir is suggested by its mechanism of action, and to date, it has demonstrated in vitro activity against the Arenaviridae, Flaviviridae, Filoviridae, Paramyxoviridae, Pneumoviridae, and Coronaviridae viral families.  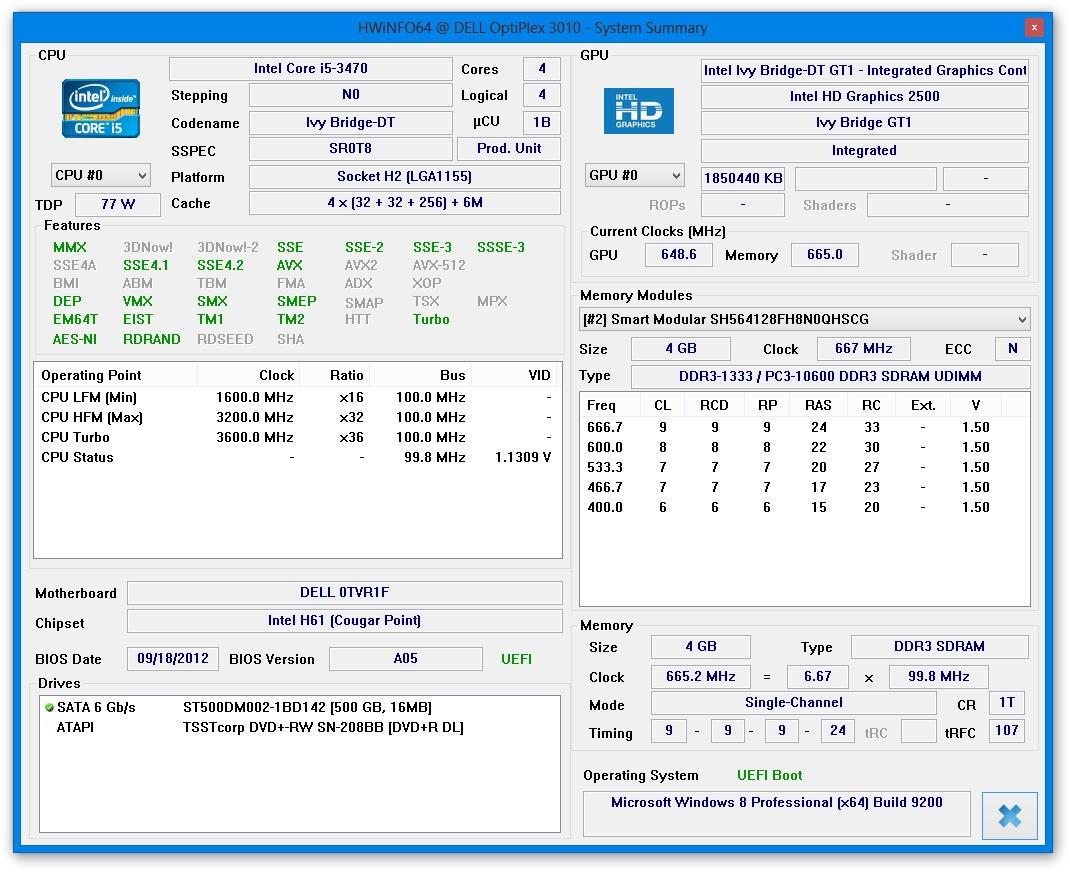
Remdesivir (GS-5734) is an adenosine triphosphate analogue first described in the literature in 2016 as a potential treatment for Ebola. Like other RNA viruses, SARS-CoV-2 depends on an RNA-dependent RNA polymerase (RdRp) enzyme complex for genomic replication, which can be inhibited by a class of drugs known as nucleoside analogues. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is the causative agent of coronavirus disease 2019 (COVID-19), which is a respiratory disease that is capable of progressing to viral pneumonia and acute respiratory distress syndrome (ARDS) COVID-19 can be fatal.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
